Control and screening of mineral oil products used in IVF fall largely on us, the manufacturer. The industry’s current approach includes indirect assays of clinical performance such as Mouse Embryo Assay (MEA) and Sperm Survival Test (SST), indirect assays of solvent properties such as pH, osmolality, and color, and semi-quantitative measures of bioburden.
However, assaying manufacturing precision offers little insight into the levels of physicochemical purity and variation present in mineral oil products used for culture overlay1,2. To remedy this we test our oil products using what we call Atomic Fingerprinting – a set of physicochemical tests to assess oil quality.
In this article, Charles Smith PhD, Director of Operations at ORIGIO, discusses the challenges faced when preparing, testing and using oil. He explains the role of Atomic Fingerprinting tests, what they involve, and how they can help offer additional assurance of high-quality products.
Despite the wide use of mineral oil for human embryo culture and micromanipulation procedures in the IVF laboratory, mineral oil is commonly considered to be one of the least defined components of the entire embryo culture system.
The human embryo culture system is a complex coordination of variables, which creates the ideal macro and micro-environment surrounding highly sensitive gametes and developing embryos.
To confidently and routinely develop viably competent embryos, this environment must be well controlled with limited stress exerted on the environment, equipment, culture media, and technical staff performance.
During in vitro fertilization, mineral oil functions as a protective barrier between the developing embryos in culture and the immediate environment.
The mineral oil overlay helps prevent evaporation of the medium and diminishes the effects of sudden fluctuations in osmolality, temperature, pH, and gas exchange.
Challenges of embryo culture with high-quality mineral oil
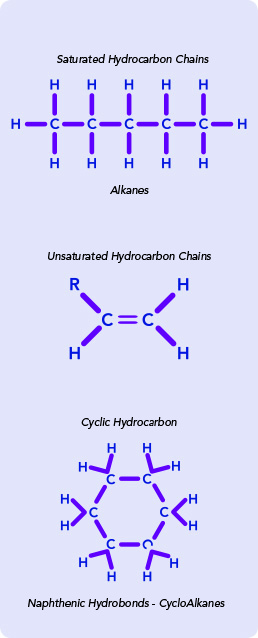
Used as an overlay, mineral oil helps maintain the thermal, osmotic and pH stability of embryos. Less refined mineral oil may be composed of a relatively larger number of unsaturated alkenes. These need to be avoided as oxidation of the alkene double bonds results in increased levels of peroxides, which are detrimental to embryos.
Variations in the physical and chemical properties of mineral oil lots can lead to inconsistent performance and possible negative clinical outcomes in the IVF laboratory3.
To find out more about sourcing and purification of oils download our white paper here
Why did we devise Atomic Fingerprinting?
Our oils are composed of stable saturated (paraffinic) hydrocarbon chains. However, in order to provide additional certainty and confidence that they are of the highest quality and stability, we have introduced Atomic Fingerprinting tests which screen for the presence of unsaturated or cyclic precursor molecules.
Atomic Fingerprinting employs a combination of advanced analytical techniques using state-of-the-art instrumentation
What is Atomic Fingerprinting?
This comprehensive and unique approach is used to systematically assess the levels of physicochemical variation in our oil products. It provides quantitative molecular data of product purity, consistency, and reliability using a variety of analytical and quantitative techniques.
Together, they help strengthen the control and screening capabilities of the oils we manufacture.
What are the tests?
Atomic Fingerprinting employs a combination of the following:
Fourier Transform Infrared Spectroscopy (FTIR)
FTIR is a robust and precise tool used for the identification and verification of chemical samples. It measures and analyzes the absorption of infrared (IR) radiation of the sample at different wavelengths4. Different chemical functional groups will produce IR spectra in specific and reproducible patterns when analyzed by FTIR. The resulting FTIR spectra can be interpreted, with additional comparisons to known chemical libraries, to provide a positive identification of each sample component and a measurement of the amount. The spectral features from the FTIR analysis should be consistent with a mixture of fully saturated hydrocarbons.
Nuclear Magnetic Resonance Spectroscopy (NMR)
NMR is a sensitive and effective method to unambiguously and quantitatively characterize a material based on the quantum mechanical magnetic property of the atoms’ nuclei. It can detect minute differences in the unique local chemical, electronic and spatial environment of an atom, allowing for the identification of particular isotopes of the chemical elements present in the sample5. This means that a carbon in a straight-chained, fully saturated alkane will register a different spectrum from a branched, unsaturated, cyclic or aromatic carbon nucleus. The result of the analysis is based on the NMR spectrum of the material, and it is detecting chemical shifts attributable to double bonds, aromatic groups or other functional groups.
The FTIR and NMR analyses confirm that our oils only contain saturated C-C bonds.
Gas Chromatography with Flame Ionization Detection (GC-FID)
This technique relies upon chromatographic separation of sample components, by virtue of their relative volatility, followed by ionization and further separation according to their molecular weight (MW). Each component is identified through matching with an extensive computer database. Hence, GC-FID can detect any trace contaminants or molecular outliers, and illustrates the MW distribution patterns of alkanes within the mineral oil.
This analysis confirms there are no volatile components with boiling point <= 250⁰C.
Kinematic Viscosity
As viscosity is an important physical property of mineral oil, we verify our oils using the kinematic viscosity method. Kinematic viscosity measurements differ from those of standard absolute viscosity measurements by taking into consideration the specific gravity of the sample. The method also measures the viscosity of the mineral oil at 40°C, which more closely emulates the temperature at which an embryologist would use equilibrated mineral oil for embryo culture or micromanipulation.
This analysis helps us ensure our oils have the right viscosity for handling.
Our ORIGIO Liquid Paraffin and SAGE Oil for Tissue Culture are both tested using Atomic Fingerprinting
Further specialized oil testing
Another more specialized test we employ for testing our oil is an Extended Mouse Embryo Assay (eMEA).
We also undertake the following well-known quality control tests:
- Mouse Embryo Assay (MEA)
- Limulus Amebocyte Lysate (LAL)
- Sterility Test
- Sperm Survival Test (SST)
- Peroxide Value Test (POV)
For further information about these tests, download our white paper here
Meeting expectations
We’re continually working hard to secure the most robust process that assures the integrity of oil batches during shipping and handling. Thorough testing of new batches (all containers within the same lot) to assure quality is also a top priority.
In addition to performing the usual industry standard tests for oil, our more specialized and sensitive tests and controls are helping provide extra scrutiny, offering a rigorous quality assessment program to deliver high-quality oil.
Further Reading: Expert Article
Scientific Director at Fertility Associates in Auckland, New Zealand, Dean Morbeck PhD, has written an article Making Sense of Oil Quality Control in the March edition of our scientific newsletter – ART Scientific.
ART Scientific is a quarterly publication that explores current topics of interest in our industry. It is written by a community of experts in the field and has a science focus at its heart.
 Charles S Smith PhD
Charles S Smith PhD
Charles Smith PhD is Director of Operations at ORIGIO. Prior to joining CooperSurgical Incorporated, he conducted research, coordinated the commercialization of new technologies, and coordinated the career development of NIH research scientists.
His research centered around the visualization and computational reconstruction of the biophysical properties that govern viral particle assembly within cells. In 2010, he was recruited from the National Institutes of Health in Bethesda, Maryland (US), and joined CooperSurgical as Director of Operations for SAGE IVF Culture Media in Pasadena, CA.
In 2015, concurrent with the consolidation of our global IVF media production, he moved to ORIGIO’s headquarters in Måløv, Denmark. In October 2016, he assumed responsibility for CooperSurgical’s global genomic laboratory and IVF media manufacturing footprint.

 My Clinic is in the United States
My Clinic is in the United States My Clinic is in Canada
My Clinic is in Canada